Daltons atomic theory4/4/2024  
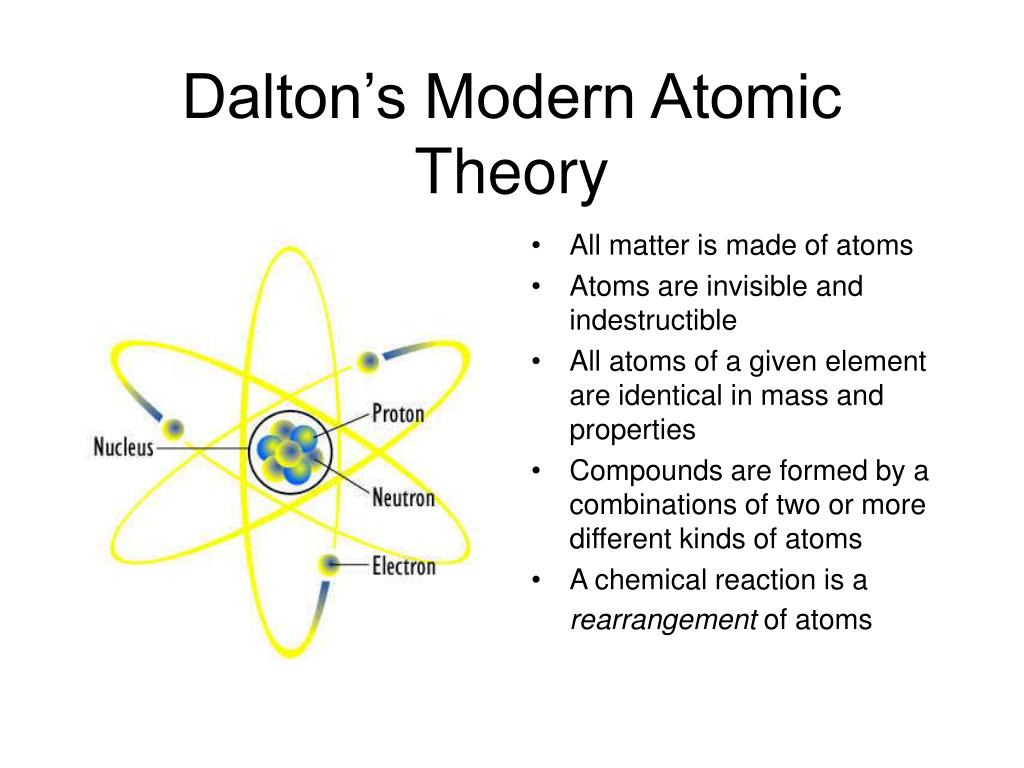
Some postulates of this theory remain valid even in today’s modern chemical thoughts. Dalton’s Atomic Theory also suggested that an atom is the smallest part of an atom that can take part in a chemical reaction.This implies that the Dalton atomic theory fails to explain the differences in properties of charcoal, graphite, and diamond (allotropes of carbon). Dalton Atomic Theory fails to explain the existence of allotropes.But this is not true in case of complex organic compounds. According to Dalton Atomic Theory, when atoms of different elements (atoms of two or more elements) combine in simple whole number ratios, we get chemical compounds.These atoms with similar atomic masses are isobars. For example, Argon (Ar) and Calcium (Ca) atoms, each have an atomic mass of 40 amu. However, this is not correct for all situations. Also, according to Dalton Atomic Theory, atoms of two-different elements differ in mass, size and many other chemical or physical properties.For example, Chlorine (Cl) has 2 isotopes with the mass numbers of 35 and 37. These atoms with the different masses are known as isotopes. But, practically we observe that atoms of several elements differ in their densities and masses. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
